Johnson & Johnson’s Acquisition of Intra-Cellular Therapies
Deal Overview
Key Facts
Acquirer: Johnson & Johnson
Target: Intra-Cellular Therapies, Inc.
Transaction value: approximately $14.6 billion
Consideration: $132.00 per share in cash
Premium: approximately 39% over Intra-Cellular's pre-announcement share price
Structure: Reverse subsidiary merger under the Delaware General Corporation Law (DGCL)
Announcement: 13 January 2025
Signing: 10 January 2025
Completion: 2 April 2025
Johnson & Johnson's acquisition of Intra-Cellular Therapies represents one of the most significant biopharmaceutical transactions of 2025. The deal reflects a broader trend in which large pharmaceutical companies acquire innovative biotechnology firms to reinforce long-term growth pipelines and mitigate revenue pressure from upcoming patent expirations. This strategic move aligns with an industry-wide shift where digital transformation and technological advancements are reshaping drug development and commercialisation strategies. By integrating Intra-Cellular's assets, Johnson & Johnson strengthens its neuroscience portfolio, particularly through CAPLYTA (lumateperone), a rapidly growing treatment for schizophrenia and bipolar depression. For Intra-Cellular shareholders, the transaction delivered a substantial premium while positioning the company's therapies within a global commercial platform capable of accelerating clinical development and international distribution.
Company Details
Acquirer - Johnson & Johnson
Founded: 1886
CEO (at time of deal): Joaquin Duato (Chairman and Chief Executive Officer)
Market capitalisation (at time of deal): approximately $370 billion
Johnson & Johnson has evolved into one of the world's largest healthcare companies. Historically operating across pharmaceuticals, medical devices and consumer products, the company sharpened its strategic focus following the 2023 separation of its consumer health division into Kenvue. This restructuring left Johnson & Johnson concentrated on two primary segments: Innovative Medicine and MedTech. The company's strategic evolution has been significantly influenced by technological advancements, including the adoption of generative AI and digital solutions across research, supply chain and clinical operations, which have enabled more efficient drug discovery and development processes.
By 2024, the Innovative Medicine segment generated approximately $57 billion in revenue, supported by several blockbuster therapies. Among its largest revenue drivers were:
DARZALEX (daratumumab) — a leading oncology treatment that has consistently demonstrated strong growth, with sales rising 23.0% year over year to $14.35 billion in 2025, reflecting its position as a cornerstone of Johnson & Johnson's oncology franchise.
STELARA (ustekinumab) — a major immunology drug facing increasing biosimilar competition following the loss of US patent exclusivity in 2025. Multiple biosimilar versions, launched by Amgen, Teva, Samsung Bioepis and others, have entered the market.
SPRAVATO (esketamine) — an innovative therapy for treatment-resistant depression that has demonstrated strong growth, with sales increasing nearly 55% in Q3 2024 alone, positioning it as a key growth driver in Johnson & Johnson's neuroscience portfolio.
Johnson & Johnson's strategic strengths lie in its global distribution capabilities, capital resources and regulatory expertise, which allow it to scale pharmaceutical innovations such as the drugs highlighted above, internationally. However, the company faces ongoing pressure from patent expirations and biosimilar competition. Stelara's loss of exclusivity negatively impacted the Innovative Medicine segment's growth by 1,110 basis points in 2025, with Stelara sales declining 41.3% to $6.08 billion. Looking ahead, Johnson & Johnson expects generic competition for Simponi and Opsumit to begin in 2026, while the company will also lose patent protection for Imbruvica around 2026. This patent cliff makes continuous pipeline development essential. The acquisition of Intra-Cellular Therapies forms part of Johnson & Johnson's broader strategy of acquiring innovative assets capable of sustaining long-term growth.
Target - Intra-Cellular Therapies
Founded: 2001
CEO (at time of deal): Dr Sharon Mates
Estimated market capitalisation (at time of deal): approximately $10 billion
Intra-Cellular Therapies was founded in 2001 and developed as a specialised biotechnology company focused on treatments for psychiatric and neurological disorders. The company built its scientific approach around small-molecule drug development targeting central nervous system conditions. Its primary commercial success was CAPLYTA (lumateperone), a first-in-class small molecule that provides selective and simultaneous modulation of serotonin, dopamine and glutamate—three neurotransmitter pathways implicated in severe mental illness. The drug received regulatory approval from the United States Food and Drug Administration for schizophrenia in 2019 and for depressive episodes associated with bipolar disorder in 2021.
CAPLYTA has experienced rapid market adoption. In 2024, the company reported $680.5 million in net product sales, representing approximately 47% year-on-year growth. CAPLYTA generated net product sales of $175.2 million in Q3 2024 alone, reflecting an increase from $125.8 million in the same quarter of 2023, with total prescriptions growing by 38% year-over-year. Analysts projected that annual sales could exceed $1 billion in the near term and potentially surpass $2.5 billion annually if additional indications receive approval. Johnson & Johnson estimates CAPLYTA has the potential for peak annual sales above $5 billion.
In the competitive landscape, CAPLYTA faces several established competitors. Johnson & Johnson itself markets Risperdal (risperidone) in the antipsychotic space. Other key competitors include AbbVie's Vraylar (cariprazine), which AbbVie's CEO has estimated has peak sales potential of $4 billion, as well as Pfizer's Geodon and established therapies such as Abilify and Latuda. Notably, recent clinical setbacks for competitors have created a favourable environment for CAPLYTA: AbbVie's acquisition of Cerevel for $8.7 billion resulted in a $3.5 billion impairment after its lead schizophrenia candidate emraclidine failed Phase II trials. This has made CAPLYTA an even more attractive strategic asset.
Beyond CAPLYTA, Intra-Cellular maintained a research pipeline targeting multiple central nervous system conditions, including major depressive disorder (for which an sNDA was submitted to the FDA in December 2024), generalised anxiety disorder (ITI-1284 in Phase II) and neurological complications associated with neurodegenerative diseases, including Alzheimer's disease-related psychosis and agitation. Although the company demonstrated strong growth potential, it also faced typical biotechnology risks such as dependence on a limited number of products and significant research costs. The acquisition allowed shareholders to realise immediate value, while transferring development and scaling risks to a global pharmaceutical organisation.
The Acquisition
Timeline
The acquisition progressed rapidly once negotiations began. The merger agreement—formally the Agreement and Plan of Merger among Johnson & Johnson, Fleming Merger Sub, Inc., and Intra-Cellular Therapies, Inc.—was signed on 10 January 2025 and publicly announced three days later.
The parties subsequently completed the regulatory and shareholder approval processes required for a United States public-company merger. The transaction closed on 2 April 2025, meaning the deal moved from announcement to completion in approximately three months. Johnson & Johnson agreed to pay $132 per share in cash, representing roughly a 39% premium over Intra-Cellular's pre-announcement share price.
Strategic Motivation
For Johnson & Johnson, the acquisition strengthens its neuroscience franchise by adding a commercial-stage therapy with significant growth potential. CAPLYTA provides both an immediate revenue stream and opportunities for additional regulatory approvals across new therapeutic indications, particularly in major depressive disorder—a condition affecting approximately 21 million adults in the United States alone. If approved for MDD, CAPLYTA has the potential to add at least $1 billion in additional peak sales. The acquisition also brings ITI-1284, a promising Phase II compound for generalised anxiety disorder and Alzheimer's-related psychosis, further deepening Johnson & Johnson's CNS pipeline.
More broadly, the acquisition helps Johnson & Johnson address longer-term revenue pressures associated with upcoming patent expirations within its pharmaceutical portfolio. Acquiring innovative biotechnology assets has therefore become a key component of the company's strategic approach to sustaining growth. The deal positions Johnson & Johnson ahead of competitors such as AbbVie, which has become more cautious in CNS investing following its Cerevel setback, and other major players like Novartis, which paid $12 billion for Avidity Biosciences to deepen its neuroscience presence.
For Intra-Cellular Therapies, the transaction delivered an attractive premium for shareholders and provided access to the global infrastructure of a major pharmaceutical company. Developing and commercialising neurological drugs is capital intensive, and integration into Johnson & Johnson's platform offers the potential to accelerate both clinical research and international distribution.
Integration
The transaction was executed using a reverse subsidiary merger, a common structure in United States public-company acquisitions. Johnson & Johnson formed a wholly owned acquisition vehicle, Fleming Merger Sub, Inc., which merged with Intra-Cellular Therapies. Following completion of the merger, the acquisition vehicle ceased to exist and Intra-Cellular survived as the legal entity, becoming a wholly owned subsidiary of Johnson & Johnson. This structure allows the target company to retain its corporate existence, including its contracts, regulatory approvals and intellectual property rights, thereby simplifying operational integration. After completion of the transaction, Johnson & Johnson indicated that Intra-Cellular would operate within its Innovative Medicine division, enabling integration into the company's global commercial infrastructure while maintaining scientific continuity.
Legal Contentions and Regulatory Impact
Delaware Merger Law
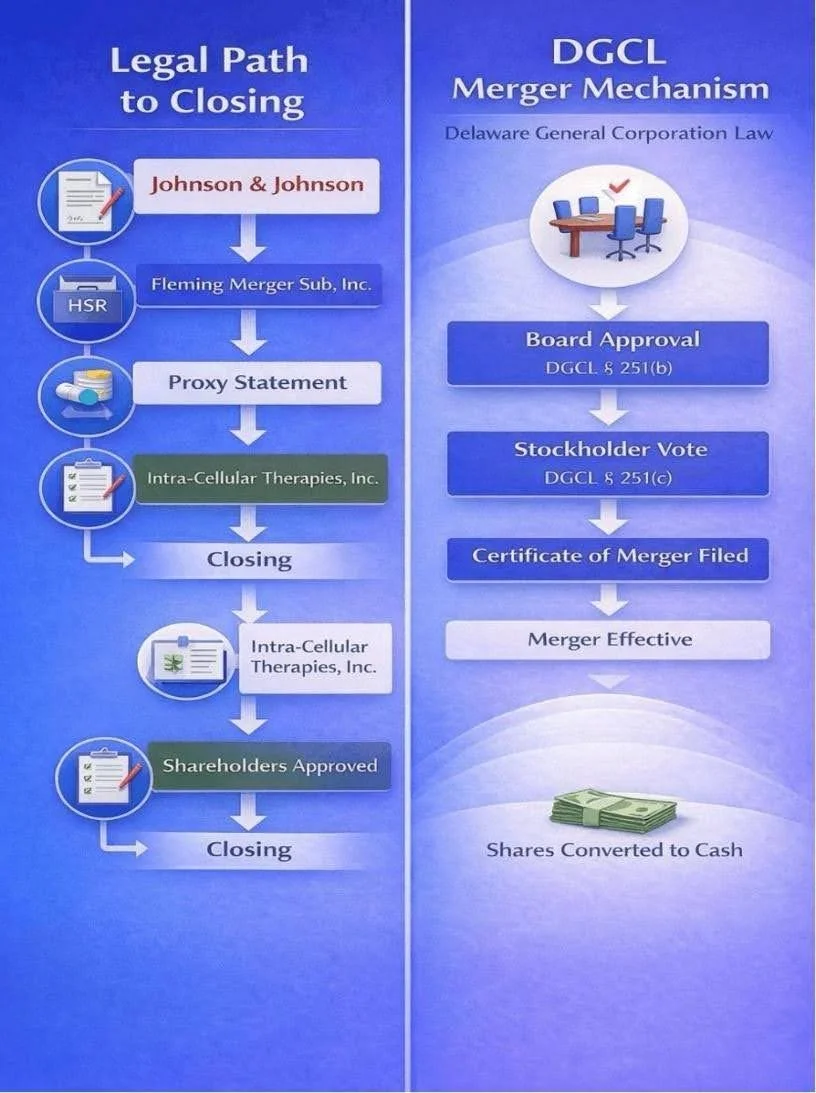
The core legal mechanism for this transaction was the Delaware General Corporation Law (DGCL). Considering Intra-Cellular Therapies is incorporated in Delaware and the merger subsidiary was also organised under Delaware law, the statutory framework governing the transaction derived from the DGCL.
The principal operative provision is Section 251, which governs mergers and consolidations of domestic corporations. Section 251(b) requires the boards of directors of each constituent corporation to approve an agreement of merger specifying the terms of the transaction and the consideration to be paid to stockholders. Following board approval, Section 251(c) requires submission of the agreement to stockholders for approval unless a statutory exception applies.
In a reverse subsidiary merger structure such as this one, the target company's i.e. Intracellular’s stockholders must approve the transaction, while the acquirer's i.e. Fleming Merger Sub, inc.’s stockholders typically do not vote because the acquirer itself is not a constituent corporation to the merger. The merger became legally effective once a certificate of merger was filed with the Delaware Secretary of State. At that moment, the merger subsidiary ceased to exist and Intra-Cellular Therapies continued as the surviving corporation, wholly owned by Johnson & Johnson.
Another relevant provision is Section 262 of the DGCL, which governs appraisal rights. Under this provision, dissenting stockholders who follow specific procedural requirements may petition the Delaware Court of Chancery to determine the "fair value" of their shares. The Figure below shows a snapshot of the legal process of the deal, illustrating the timeline from board approval, stockholder vote, and regulatory clearance to the filing of the certificate of merger with the Delaware Secretary of State.
Hart-Scott-Rodino Antitrust Review
The second key legal framework governing the transaction was the Hart-Scott-Rodino Antitrust Improvements Act of 1976. Under this regime, parties to large mergers must file pre-merger notification with the Federal Trade Commission (FTC) and the Department of Justice Antitrust Division and observe a waiting period before closing the transaction.
The purpose of the HSR regime is to allow regulators to determine whether a proposed acquisition could substantially lessen competition such as resulting in monopoly in antipsychotic drug market, unfettered control over pricing and entry of new competitors, under Section 7 of the Clayton Act. In assessing the transaction, regulators would have considered whether Johnson & Johnson's acquisition of Intra-Cellular Therapies could create too dominant a market position in the antipsychotic drug market, potentially affecting pricing, distribution and innovation. Given Johnson & Johnson's existing presence in the CNS space through Risperdal and Invega Sustenna (for which patent protection was recently upheld until 2031), and the presence of strong competitors such as AbbVie (Vraylar) and Pfizer (Geodon), the transaction did not raise significant horizontal concentration concerns. The waiting period expired without the issuance of a Second Request, indicating that regulators did not identify significant competition concerns.
Securities Disclosure and Stockholder Approval
As Intra-Cellular Therapies was a publicly traded company listed on the Nasdaq Global Select Market, its stockholders were required to receive a proxy statement containing detailed disclosure about the transaction under Schedule 14A of the Securities Exchange Act of 1934. The proxy statement described the background of the merger, the board's recommendation, the fairness opinion of the company's financial adviser, and the interests of directors and executive officers in the transaction. Stockholders approved the merger agreement at a special meeting held on 27 March 2025, clearing the final procedural step before closing.
Industry Impact
The transaction highlights an important trend in the pharmaceutical industry: large companies are increasingly acquiring biotechnology firms possessing commercial-stage therapies or promising late-stage pipelines. The neurodegenerative disease and psychiatric disorders spaces are particularly attractive due to the large patient population and historically limited innovation. Indeed, record acquisitions in both 2024 and 2025 were driven by neuroscience-focused biotechs, and J&J's $14.6 billion acquisition of Intra-Cellular Therapies was the largest M&A deal of 2025. By October 2025, the industry had reached an aggregate takeover value of $70 billion, with 2025 significantly outperforming the previous year in deal value. CAPLYTA's growth trajectory and potential additional indications made Intra-Cellular Therapies an attractive strategic target. The acquisition reinforces the competitive race among large pharmaceutical companies to secure differentiated assets capable of supporting future revenue growth, particularly as the industry navigates an unprecedented patent cliff affecting multiple blockbuster drugs across major players.
House View
From a strategic perspective, the acquisition appears coherent. Johnson & Johnson required future revenue growth for which it requires new drugs and constant innovation which is synergized by this acquisition for neuroscience portfolio and greater depth in neuroscience therapeutics, while Intra-Cellular Therapies offered both an established commercial asset and a promising development pipeline. The timing is particularly shrewd: with CAPLYTA's patent protection extending until 2033 and the drug poised for potential FDA approval in major depressive disorder, Johnson & Johnson gains a durable revenue stream just as its legacy neuroscience asset Invega Sustenna faces patent pressures. The deal also capitalises on the favourable industry environment following competitor setbacks, positioning Johnson & Johnson to capture additional market share in the CNS space.
In our view, provided CAPLYTA continues its commercial trajectory and additional indications are successfully developed, this acquisition may prove to be a well-timed example of how large pharmaceutical companies can use targeted acquisitions to strengthen therapeutic franchises before major patent expirations occur. However, we believe competition in the CNS drug market arises suddenly from scientific breakthroughs—as illustrated by AbbVie’s $3.5 billion impairment after its lead schizophrenia candidate failed Phase II trials, and by the rapid emergence of Chinese and Korean biotechs challenging established patent fortresses. Therefore, it will be important for Johnson & Johnson to place paramount focus on being a proactive torchbearer of new discoveries by utilising Intra-Cellular’s specialised R&D strengths, rather than responding reactively to market disruptions; preserving its scientific agility and internal innovation engine will be critical to sustaining long-term advantage in a field where clinical setbacks can reshape the competitive landscape overnight.
References
Deal Overview / Transaction Details
1. Johnson & Johnson, 'Johnson & Johnson Strengthens Neuroscience Leadership with Acquisition of Intra-Cellular Therapies, Inc.' (Press Release, 13 January 2025) [https://www.investor.jnj.com/news-details/2025/Johnson--Johnson-Strengthens-Neuroscience-Leadership-with-Acquisition-of-Intra-Cellular-Therapies-Inc/default.aspx](https://www.investor.johnsonandjohnson.com/news-releases/news-release-details/johnson-johnson-strengthens-neuroscience-leadership-acquisition )
2. Johnson & Johnson, 'Johnson & Johnson Completes Acquisition of Intra-Cellular Therapies, Inc.' (Press Release, 2 April 2025) [https://markets.businessinsider.com/news/stocks/johnson-johnson-completes-intra-cellular-therapies-acquisition-sees-dilution-1034553213](https://markets.businessinsider.com/news/stocks/johnson-johnson-completes-intra-cellular-therapies-acquisition-sees-dilution-1034553213 )
3. Intra-Cellular Therapies Inc, Current Report (Form 8-K) (13 January 2025) US Securities and Exchange Commission [https://ir.intracellulartherapies.com/sec-filings/all-sec-filings/content/0001193125-25-004052/0001193125-25-004052.pdf](https://ir.intracellulartherapies.com/sec-filings/all-sec-filings/content/0001193125-25-004052/0001193125-25-004052.pdf )
4. Reuters, 'Johnson & Johnson to buy Intra-Cellular Therapies for $14.6 billion' (13 January 2025) [https://www.reuters.com/business/healthcare-pharmaceuticals/jj-buy-intra-cellular-therapies-146-billion-2025-01-13/](https://www.reuters.com/business/healthcare-pharmaceuticals/jj-buy-intra-cellular-therapies-146-billion-2025-01-13/ )
Johnson & Johnson Company Information
5. Johnson & Johnson, Annual Report 2024 (Form 10-K) US Securities and Exchange Commission [https://d18rn0p25nwr6d.cloudfront.net/CIK-0000200406/5351612d-a918-4f2e-928a-d20e06dbcf45.pdf](https://d18rn0p25nwr6d.cloudfront.net/CIK-0000200406/5351612d-a918-4f2e-928a-d20e06dbcf45.pdf )
6. Johnson & Johnson, '2024 Annual Report' (PDF) [https://s203.q4cdn.com/636242992/files/doc_downloads/Annual_meeting/2025/Johnson-Johnson-2024-Annual-Report.pdf](https://s203.q4cdn.com/636242992/files/doc_downloads/Annual_meeting/2025/Johnson-Johnson-2024-Annual-Report.pdf ) (from the original article)
7. Reuters, 'J&J beats quarterly estimates as cancer drug Darzalex drives growth' (2025) [https://www.reuters.com/business/healthcare-pharmaceuticals/johnson-johnson-beats-quarterly-profit-estimates-strong-darzalex-sales-2025-07-16/](https://www.reuters.com/business/healthcare-pharmaceuticals/johnson-johnson-beats-quarterly-profit-estimates-strong-darzalex-sales-2025-07-16/ )
8. Johnson & Johnson, Company Profile and Corporate Strategy [https://www.britannica.com/money/Johnson-Johnson](https://www.britannica.com/money/Johnson-Johnson )
Intra-Cellular Therapies Company Information
9. Intra-Cellular Therapies Inc, Annual Report (Form 10-K) 2024 US Securities and Exchange Commission [https://companiesmarketcap.com/intra-cellular-therapies/annual-reports/10-k-2024-69503/](https://companiesmarketcap.com/intra-cellular-therapies/annual-reports/10-k-2024-69503/ )
10. Intra-Cellular Therapies Inc, Quarterly Report (Form 10-Q) US Securities and Exchange Commission [https://www.sec.gov/Archives/edgar/data/1567514/000162828024044196/0001628280-24-044196-index.htm](https://www.sec.gov/Archives/edgar/data/1567514/000162828024044196/0001628280-24-044196-index.htm )
11. AP News, 'Johnson & Johnson to buy Intra-Cellular Therapies for $14.6 billion' (2025) [https://apnews.com/article/johnson-johnson-intracellular-therapies-jnj-buying-fb32e72a1e44831b69db768cb48c3b0a](https://apnews.com/article/johnson-johnson-intracellular-therapies-jnj-buying-fb32e72a1e44831b69db768cb48c3b0a )
12. U.S. Food and Drug Administration, CAPLYTA (lumateperone) Approval Information
[https://www.fda.gov/drugs](https://www.fda.gov/drugs ) (from the original article)
Deal Structure / Integration
13. Intra-Cellular Therapies Inc,Definitive Proxy Statement relating to merger with Johnson & Johnson US Securities and Exchange Commission [https://ir.intracellulartherapies.com/sec-filings/all-sec-filings/content/0001193125-25-043441/0001193125-25-043441.pdf](https://ir.intracellulartherapies.com/sec-filings/all-sec-filings/content/0001193125-25-043441/0001193125-25-043441.pdf )
14. Cravath, Swaine & Moore LLP, 'Cravath Represents Johnson & Johnson in Acquisition of Intra-Cellular Therapies' [https://www.cravath.com/news/LMG-Life-Sciences-Honors-Cravath-with-2025-Impact-Deal-Award.html](https://www.cravath.com/news/LMG-Life-Sciences-Honors-Cravath-with-2025-Impact-Deal-Award.html )
15. Wachtell, Lipton, Rosen & Katz, Client Matters: Intra-Cellular Therapies Acquisition [https://www.wlrk.com](https://www.wlrk.com ) (from the original article)
Delaware Merger Law
16. Delaware General Corporation Law, 8 Del. C. § 251 (Merger or Consolidation of Domestic Corporations) [https://delcode.delaware.gov/title8/c001/sc09/index.html](https://delcode.delaware.gov/title8/c001/sc09/index.html ) (from the original article)
17. Delaware General Corporation Law, 8 Del. C. § 262 (Appraisal Rights) [https://www.sec.gov/Archives/edgar/data/1001836/000119312515363939/d99047dex1082.htm](https://www.sec.gov/Archives/edgar/data/1001836/000119312515363939/d99047dex1082.htm )
18. Lawrence A Hamermesh and Leo E Strine Jr, 'Delaware Corporate Law and the Role of the Board in Mergers' (Harvard Law School Forum on Corporate Governance) [https://corpgov.law.harvard.edu/](https://corpgov.law.harvard.edu/ ) *(from the original article)
Antitrust Review (HSR)
19. Hart-Scott-Rodino Antitrust Improvements Act of 1976, 15 USC § 18a [https://www.ftc.gov/legal-library/browse/statutes/hart-scott-rodino-antitrust-improvements-act-1976](https://www.ftc.gov/legal-library/browse/statutes/hart-scott-rodino-antitrust-improvements-act-1976 )
20. Clayton Act 1914, Section 7 (15 USC § 18) [https://www.law.cornell.edu/uscode/text/15/18](https://www.law.cornell.edu/uscode/text/15/18 )
21. Federal Trade Commission, 'Premerger Notification and the Hart-Scott-Rodino Act' [https://www.ftc.gov/enforcement/premerger-notification-program](https://www.ftc.gov/enforcement/premerger-notification-program ) *(from the original article)
Securities Disclosure and Shareholder Approval
22. Securities Exchange Act of 1934, Schedule 14A Proxy Disclosure Requirements [https://www.ecfr.gov/current/title-17/section-240.14a-101 ](https://www.ecfr.gov/current/title-17/section-240.14a-101 )
23. U.S. Securities and Exchange Commission, 'Proxy Rules and Schedule 14A' [https://www.sec.gov/rules-regulations/proxy-rules](https://www.sec.gov/rules-regulations/proxy-rules ) (from the original article)
Industry Context
24. Reuters, 'Big Pharma accelerates biotech dealmaking amid patent cliff pressures' (2025) [https://www.reuters.com/business/healthcare-pharmaceuticals/big-pharma-accelerates-biotech-dealmaking-amid-patent-cliff-pressures-2025-10-27/], (https://www.reuters.com/business/healthcare-pharmaceuticals/big-pharma-accelerates-biotech-dealmaking-amid-patent-cliff-pressures-2025-10-27/ )
25. Financial Times, 'Drugmakers turn to acquisitions to replenish pipelines' (2024) [https://www.channelchek.com/articles/industry-news/healthcare/big-pharma-goes-bio-prospecting-why-major-drug-makers-are-buying-innovative-biotech-startups-36638](https://www.channelchek.com/articles/industry-news/healthcare/big-pharma-goes-bio-prospecting-why-major-drug-makers-are-buying-innovative-biotech-startups-36638 )